
Recently, scientist from SHVRI CAAS found that RNASEK interacted with PEDV structural proteins facilitates virus entry via clathrin-mediated endocytosis.
Background
Porcine epidemic diarrhea virus (PEDV), as a type of Alphacoronavirus causing acute diarrhea and high death rate among sucking piglets, poses great financial damage to the swine industry. Nevertheless, the molecular mechanism whereby PEDV enters host cells is unclear, limiting the development of PED vaccines and anti-PEDV agents.
Research Progress
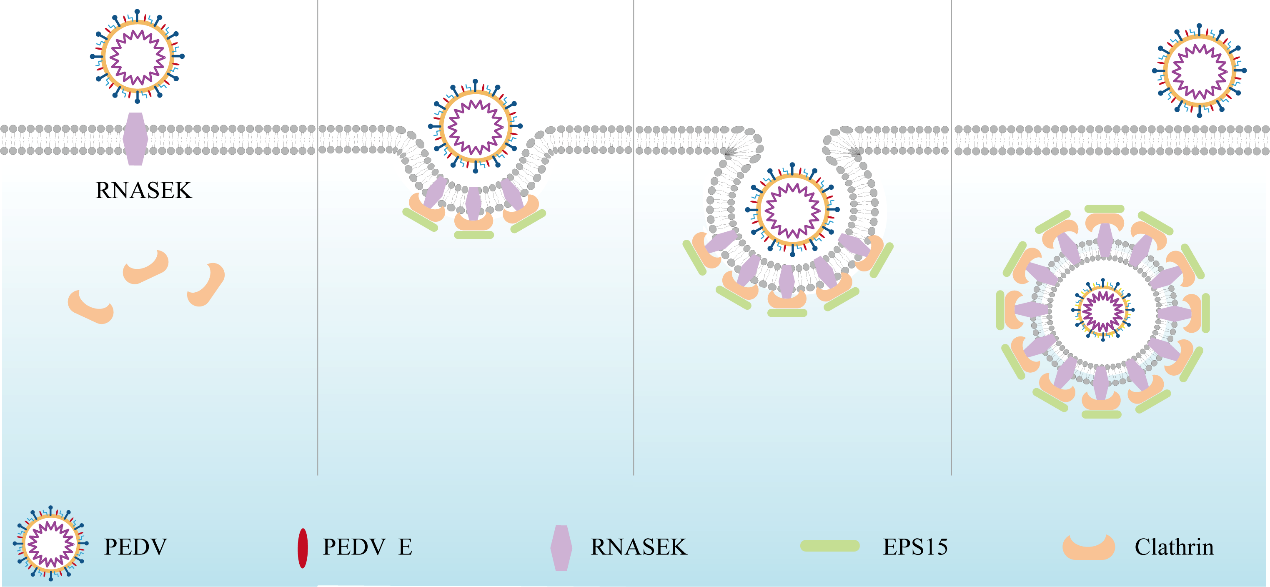
Present study found that the host protein ribonuclease kappa (RNASEK) was regulated by USF2, a transcription factor, and facilitated the PEDV replication. RNASEK was identified as a novel binding partner of PEDV, which interacted with spike (S), envelope (E), membrane (M) proteins on PEDV virion surfaces to increase the uptake not for attachment of PEDV virions. PEDV enters cells through the endocytosis pathways. RNASEK knockdown or RNASEK knockout assay revealed that through clathrin-mediated endocytosis (CME), RNASEK promoted the internalization of PEDV virions. Clathrin and the adaptor protein EPS15 only interacted with PEDV E protein, demonstrating that the RNASEK could targeting more virions through interacted with PEDV S, E, M proteins to clathrin and EPS15 proteins rather than merely interacting with PEDV E protein to mediate the PEDV entry through CME. Moreover, our findings suggest that RNASEK, a newly identified host-entry factor, facilitates PEDV internalization by increasing the interaction of PEDV virions and EPS15-clathrin complex and may also provide a potential target for anti-PEDV therapies.
Funding
This study was supported by the National Key Research and Development Program of China, National Natural Science Foundation of China, and Natural Science Foundation of Shanghai.
The Original Link
https://pubmed.ncbi.nlm.nih.gov/39835814/.

